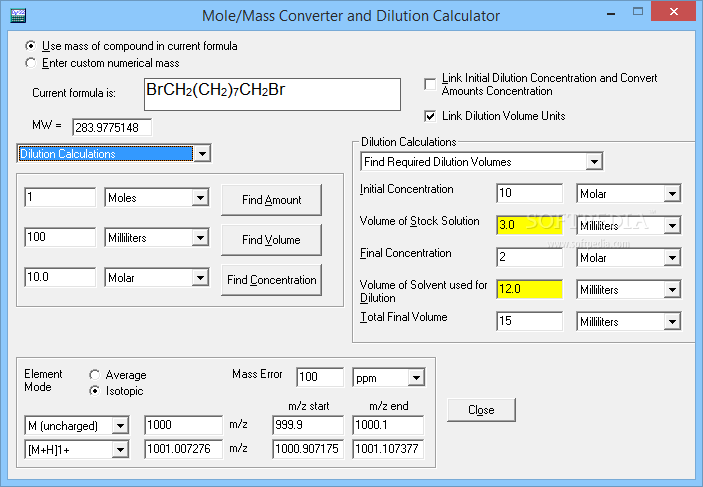
Enter 10 into the Volume box and select the correct unit (milliliter).Enter 10 into the Concentration box and select the correct unit (millimolar).Enter 197.13 into the Molecular Weight (MW) box.What is the mass of compound required to make a 10 mM stock solution in 10 ml of water given that the molecular weight of the compound is 197.13 g/mol? Mass (g) = Concentration (mol/L) x Volume (L) x Molecular Weight (g/mol) An example of a molarity calculation using the Tocris molarity calculator 
The Tocris molarity calculator is based on the following equation: *When preparing stock solutions always use the batch-specific molecular weight of the product found on the vial label and CoA (available online).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
